FOOD
Microbial detection
for food & drinks


Nissui Pharma Solutions (NPS) was founded in April 2019 as a subsidiary of Nissui Pharmaceutical Co. Ltd. Following the 2022 acquisition of our parent company Nissui Pharmaceutical by the Shimadzu Group, NPS became Shimadzu Diagnostics Europe (SDE).
SDE commercializes microbial contamination tests for the food, beverage, and pharmaceutical industries in Europe, Middle East, and Africa (EMEA).
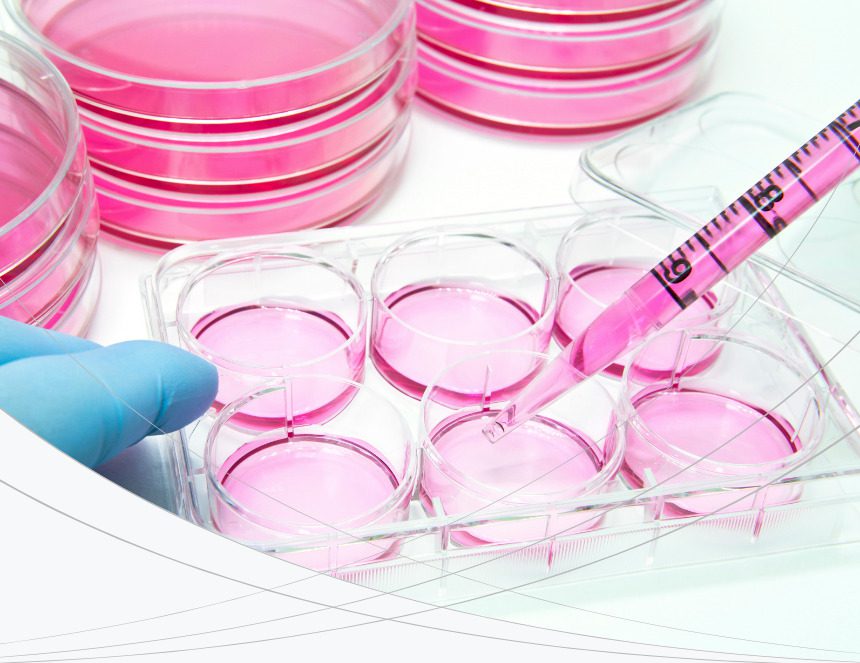
This includes CompactDry microbiology testing (and related products) to offer a range of bacterial detection solutions for the food and beverage industries. For the biopharmaceutical industry, we provide molecular biology tools for mycoplasma and sterility testing used in quality control for research, development, and production of Advanced Therapy Medicines (ATMP).
We also provide molecular biology tools for mycoplasma and sterility testing used in quality control for research, development, and production of biopharmaceuticals and Advanced Therapy Medicinal Products (ATMP).
Shimadzu Diagnostics Europe provides analytical solutions for microbiological quality control by developing high performance DNA extraction and PCR technologies.
Our solutions offer rapid and sensitive detection of microbial contaminants in line with the strict safety standards of the International Pharmacopeias and meet the challenging time constrains required to take ATMPs from production to patient.
This includes CompactDry microbiology testing (and related products) to offer a range of bacterial detection solutions for the food and beverage industries.
Learn more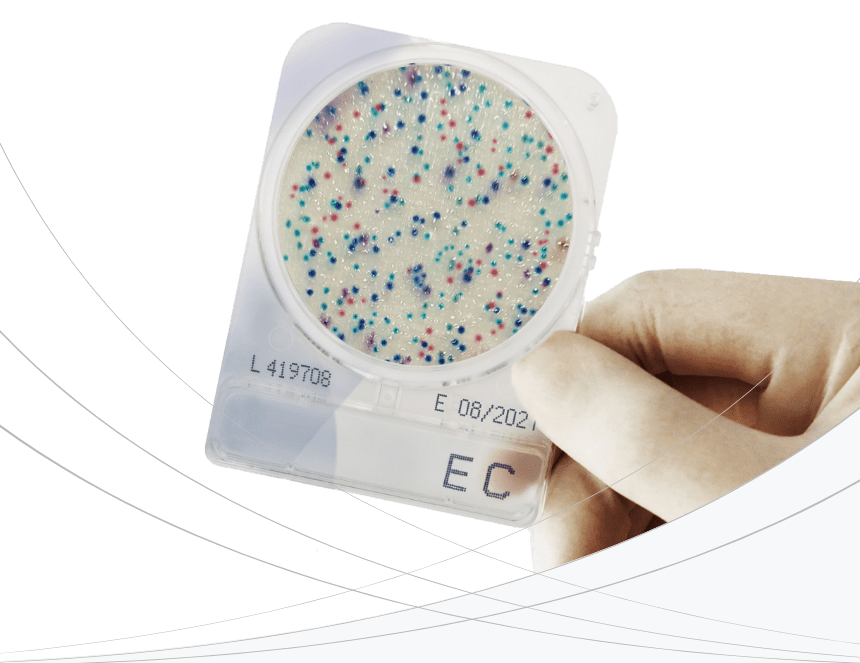
Nissui Pharmaceutical (now Shimadzu Diagnostics Corporation) originated from a major Japanese company in the fish processing industry, they had the mission to develop highly innovative solutions for the food industry.
Our products provide simple and rapid alternatives to replace traditional microbiology testing. This includes ready-to-use dehydrated, chromogenic selective media plates, colorimetric/fluorescence pathogen test for water, and an extensive range of high-quality culture media products.
Learn moreOur technical experts are always on hand to provide help, advice, and training
for all our food and pharmaceuticals contamination detection kits.
Don’t hesitate to contact us, just click below
or send an email to support@diagnostics-eu.shimadzu.com
Order our products directly through our website or by email at
orders@diagnostics-eu.shimadzu.com
Certificates of analysis are available on request, Please contact
support@diagnostics-eu.shimadzu.com
Fast delivery throughout
the EMEA guarantee
by our logistic partner in Netherlands
International certification for Food and Pharma products available on request:
support@diagnostics-eu.shimadzu.com